- WHAT IS PGT-A +
- BENEFITS OF PGT-A +
- Indications for PGT-A +
- Technology +
- Methodology +
- Mosaicism +
- Downloads +
- References +
Preimplantation Genetic Testing for Aneuploidy (PGT-A), also known as PGS (Preimplantation Genetic Screening), is used by fertility clinics across the world as an additional tool for embryo selection.
The aim of PGT-A is to genetically screen embryos to find those most likely to have the correct number of chromosomes, so that these euploid embryos are preferentially selected for transfer.
Since miscarriage rates increase exponentially beyond the age of 35, PGT-A is widely accepted as a means of identifying euploid embryos for women of advanced maternal age, to significantly improve their IVF outcome1.
PGT-A is also particularly useful in IVF programs working towards single embryo transfers (SETs)2,3. One of the current main goals of IVF worldwide is to move towards SET to minimise multiple pregnancies and their associated risks.
All couples undergoing IVF may be counselled and offered PGT-A as a means of embryo selection.
This is particularly applicable for those couples with a large number of good quality embryos (based on morphology) available for transfer.
PGT-A should be strongly considered for the following indications6,7:
- Advanced maternal age (AMA)
- Recurrent implantation failure (RIF)
- Severe male factor (SMF) infertility
- Recurrent miscarriage (RM) in couples with normal karyotypes
At Next Biosciences, we make use of the Vitrolife EmbryoMap solution on the Illumina MiSeq instrument.
The Vitrolife EmbryoMap solution screens all 24 chromosomes in the human genome using next generation sequencing (NGS) technology to detect large chromosomal copy number variations (CNV) greater than 10Mbp across the whole genome, thereby providing an accurate and efficient view of the number of chromosomes in an embryo.
The Vitrolife EmbryoMap will be replacing the well-respected VeriSeq PGS solution internationally. The Vitrolife EmbryoMap solution has also undergone extensive validation studies and has shown to sensitivities and specificities both of 99.9%8.
The technology we use can detect:
- Whole chromosomal gains or losses (aneuploidies such as trisomy, tetrasomy, monosomy, nullisomy).
- Partial chromosomal gains or losses (duplications, deletions, insertions, unbalanced translocations).
- Certain forms of polyploidy (entire additional set(s) of chromosomes).
The technology we use cannot detect:
- All forms of polyploidy.
- Balanced chromosomal rearrangements (translocations or inversions).
- Small CNV (microdeletions, microduplications, single nucleotide mutations).

Mosaicism refers to two or more cell line populations with different chromosomal complements being present within the same embryo.
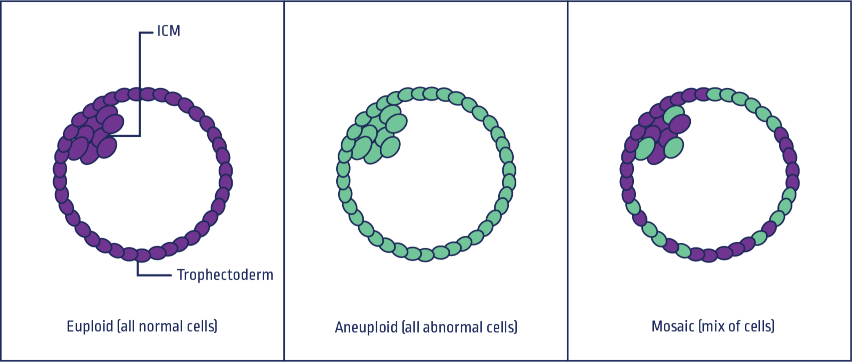
Sensitive technologies such as next generation sequencing can distinguish uniform aneuploidies from mosaic aneuploidies and can quantify the extent of the mosaicism6.
In the Next Biosciences laboratory, we report on mosaicism levels as:
- Low level mosaic: 20-50% of cells contain the abnormality detected
- High level mosaic: >50-80% of cells contain the abnormality detected
The cut-offs between high- and low- level mosaicism as above are in keeping with current international guidelines9,10 and as new data becomes available these cut-offs may be reviewed and adjusted.
Mosaic embryos can be further categorised as either whole chromosome mosaics, or segmental mosaics, where a piece of a chromosome is either extra or missing, rather than a whole chromosome.
The latest research shows that some mosaic embryos can implant and result in an ongoing pregnancy and healthy live birth, but at lower success rates compared to euploids13,14. In general, low level mosaics have a better reproductive outcome compared to high level mosaics. One multicentre study showed that segmental mosaics, irrespective of the level of mosaicism, have the best reproductive outcome compared to whole chromosome mosaics11. It is, therefore, important that your PGT-A test of choice is validated to correctly identify both low level and segmental mosaics, the VeriSeq PGS Solution which is the precursor for the current Vitrolife EmbryoMap Solution utilised in the Next Biosciences laboratory has been shown to be superior in this regard13.
Studies have shown that mosaic embryos are less likely to implant and some have also demonstrated that mosaics can result in higher miscarriage rates compared to euploid embryos13. Therefore, transfer of euploid blastocysts should always be prioritised over mosaic embryos, as euploid embryos have the best reproductive outcome9,10.
Should transfer of a mosaic embryo be considered, this should be done together with a genetic counsellor whom has experience in this field. Next Biosciences has an in-house genetic counsellor who is able to support you and any of your patients whom may need to consider a mosaic embryo transfer.
- Rubio C, Bellver J, Rodrigo L, et al. In vitro fertilization with preimplantation genetic diagnosis for aneuploidies in advanced maternal age: a randomized, controlled study. Fertil Steril. 2017;107(5):1122-1129. doi:10.1016/j.fertnstert.2017.03.011.
- Anderson RE, Whitney JB, Schiewe MC. Clinical benefits of preimplantation genetic testing for aneuploidy (PGT-A) for all in vitro fertilization treatment cycles. Eur J Med Genet. 2020;63(2):103731. doi:10.1016/j.ejmg.2019.103731.
- Gorodeckaja J, Neumann S, McCollin A, et al. High implantation and clinical pregnancy rates with single vitrified-warmed blastocyst transfer and optional aneuploidy testing for all patients [published online ahead of print, 2019 Jan 7]. Hum Fertil (Camb). 2019;1-12. doi:10.1080/14647273.2018.1551628.
- Tiegs AW, Tao X, Zhan Y, et al. A multicenter, prospective, blinded, nonselection study evaluating the predictive value of an aneuploid diagnosis using a targeted next-generation sequencing-based preimplantation genetic testing for aneuploidy assay and impact of biopsy [published online ahead of print, 2020 Aug 27]. Fertil Steril. 2020;S0015-0282(20)30711-1. doi:10.1016/j.fertnstert.2020.07.052.
- Munné S, Kaplan B, Frattarelli JL, et al. Preimplantation genetic testing for aneuploidy versus morphology as selection criteria for single frozen-thawed embryo transfer in good-prognosis patients: a multicenter randomized clinical trial. Fertil Steril. 2019;112(6):1071-1079.e7. doi:10.1016/j.fertnstert.2019.07.1346.
- ESHRE PGT Consortium Steering Committee, Carvalho F, Coonen E, et al. ESHRE PGT Consortium good practice recommendations for the organisation of PGT. Hum Reprod Open. 2020;2020(3):hoaa021. Published 2020 May 29. doi:10.1093/hropen/hoaa021.
- Practice Committees of the American Society for Reproductive Medicine and the Society for Assisted Reproductive Technology. The use of preimplantation genetic testing for aneuploidy (PGT-A): a committee opinion. Fertil Steril. 2018;109(3):429-436. doi:10.1016/j.fertnstert.2018.01.002.
- https://www.vitrolife.com/globalassets/support-documents/tech-notes/technote_validation-of-the-veriseq-pgs-solution_jan-2019.pdf
- Leigh D, Cram DS, Rechitsky S, et al. PGDIS position statement on the transfer of mosaic embryos 2021. Reprod Biomed Online. 2022 Jul;45(1):19-25.doi: 10.1016/j.rbmo.2022.03.013. Epub 2022 Mar 20. PMID: 35523707.
- ESHRE Working Group on Chromosomal Mosaicism. ESHRE survey results and good practice recommendations on managing chromosomal mosaicism. Hum Reprod Open. 2022 Nov 7;2022(4):hoac044. doi: 10.1093/hropen/hoac044. PMID: 36349144; PMCID: PMC9637425.
- Viotti M, et al. Using outcome data from one thousand mosaic embryo transfers to formulate an embryo ranking system for clinical use. Fertil Steril. 2021 May;115(5):1212-1224. doi: 10.1016/j.fertnstert.2020.11.041. Epub 2021 Mar 6. PMID: 33685629.
- Capalbo A, et al. Mosaic human preimplantation embryos and their developmental potential in a prospective, non-selection clinical trial. Am J Hum Genet. 2021 Nov 11:S0002-9297(21)00412-2. doi: 10.1016/j.ajhg.2021.11.002. Epub ahead of print. PMID: 34798051.
- Biricik A, et al. Cross-Validation of Next-Generation Sequencing Technologies for Diagnosis of Chromosomal Mosaicism and Segmental Aneuploidies in Preimplantation Embryos Model. Life (Basel). 2021 Apr 12;11(4):340. doi: 10.3390/life11040340. PMID: 33921258; PMCID: PMC8069536.
Next Biosciences
Ariane Avenue
International Business Gateway
Cnr. New Road and 6th Road
Midrand, South Africa
Office Hours
Monday - Thursday: 08h00 - 17h00
Friday: 08h00 - 16h00

If a mosaic embryo is transferred and a pregnancy is achieved, prenatal diagnosis should be discussed with a clinician with a good understanding of the benefits and limitations of all available options. While Non-Invasive Prenatal Testing (NIPT) may provide information early in a pregnancy (from 10 weeks), preference should be given to 24-chromosome NIPT methodology that includes the mosaic chromosome in question. It is important to understand that NIPT can only assess placental chromosome status. Amniocentesis is considered to be the most representative of the chromosomal complement of the fetus. All testing options should be discussed on a case-by-case basis.