A personal view from Dr Chris Vorster, Chemical Pathologist and Head: Centre for Human Metabolomics, North-West University
Newborn screening is a worldwide recognised public health measure aimed at the early screening, diagnosis and management of selected inherited diseases. Diseases are selected based on the current ability to detect and treat them and the demonstration of the positive economic impact of such interventions. Newborn screening is perhaps the pinnacle of preventative medicine. Early diagnosis and treatment ensure optimal living and allow an individual to contribute to society as opposed to becoming a long-term societal responsibility.
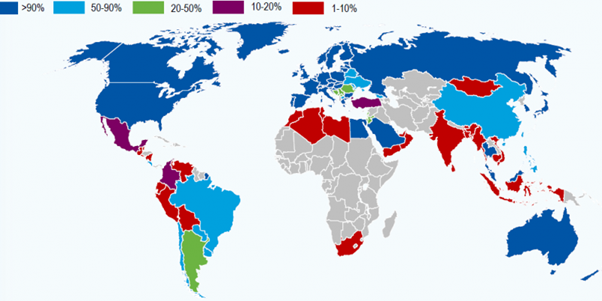
Global Distribution of Newborn Screening (2016)
The incidence of inherited metabolic diseases in South Africa
There is a perception amongst many South African clinicians that inherited metabolic diseases are exceedingly rare in our country. These perceptions are based on a lack of recognition of these conditions when they present and also on misleading earlier research.¹ In more recent research, Van der Watt et al., predicted an incidence of 1 in 5 000 newborns for type I glutaric acidemia in a selected South African black population by estimating of the carrier frequency of a commonly occurring mutation.² Prof LJ Mienie has headed the Metabolic Laboratory on the Potchefstroom Campus of North West University for the past 30 years and has received more than 50 000 requests for the metabolic workup of patients. In his experience the most frequently occurring metabolic diseases that are also included in the newborn screening panel are isovaleric acidemia, propionic acidemia, galactosaemia, vitamin B-responsive methylmalonic acidemia, maple syrup urine disease and glutaric acidemia type I. The March of Dimes global report on birth defects found that genetic and congenital disorders cut across all nations with little regard for ethnic background and socioeconomic status. There is little evidence to suggest that South Africa is spared from metabolic diseases.
Economic considerations of a newborn screening program
One of the common constraints to the implementation of a newborn screening program is the perception such a program will come a huge expense and will divert funds from competing health priorities. The reality is that ample evidence exists to illustrate that the economic benefit of screening offsets the costs and that it is economically far better to screen and initiate early treatment than to diagnose late and deal with the associated morbidity and mortality.³˒⁴˒⁵˒⁶˒⁷
While most healthcare funders will agree that preventative health initiatives will lower the healthcare expenses of an individual over their lifetime, there seems to be less clarity on how preventative programs are prioritised. In South Africa, screening for HIV, hypercholesterolemia, breast cancer, and prostate cancer is a fairly common practices. It is uncertain why these conditions were prioritised before newborn screening when at least some evidence indicates the opposite.⁷
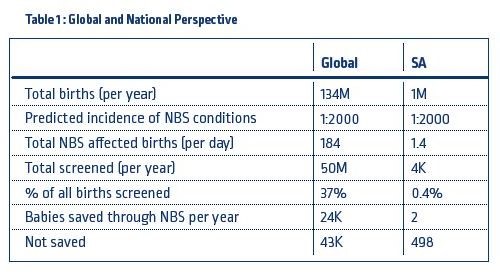
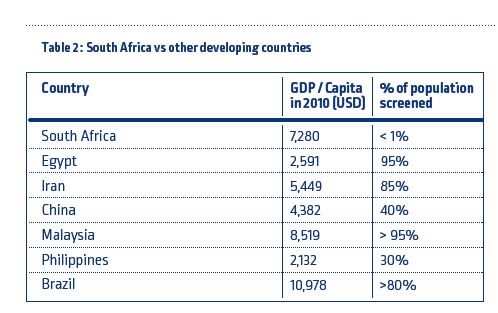
A more realistic economic barrier is that the initial cost of screening is incurred straight away while the benefits are only realised over an extended period. In light of this, most developing countries have opted for an incremental approach to implementation through pilot programs. In addition, a country must have reached a certain level of economic prosperity to make implementing a screening program feasible. Table 2 compares the economic prosperity (expressed as the GDP per Capita) and the percentage of annual newborns screened for selected developing nations.
Conclusion
There is an overwhelming case for newborn screening in South Africa. For many diseases in the newborn screening profile, simple and cost-effective treatments are available. It seems completely unethical to deny the children of South Africa such interventions especially given the arguments that are put forward in this document. The technology and know-how to perform newborn screening is available, while the logistical infrastructure already exists in many instances. The issue cannot be ignored any longer.
References
Hitzeroth H, Niehaus C, Brill D. Phenylketonuria in South Africa. SAMJ [Internet]. 1995 [cited 2013 Apr 23];85(1):33–6. Available from: http://archive.samj.org.za/1995 VOL 85 Jan-Dec/Articles/01 January/1.9 PHENYLKETONURIA IN SOUTH AFRICA.A REPORT ON THE STATUS QUO. H.W.Hitzeroth, C.E.Niehaus, D.C.B.pdf
Van der Watt G, Owen EP, Berman P, Meldau S, Watermeyer N, Olpin SE, et al. Glutaric aciduria type 1 in South Africa-high incidence of glutaryl-CoA dehydrogenase deficiency in black South Africans. Mol Genet Metab [Internet]. Elsevier Inc.; 2010 [cited 2013 Feb 4];101(2-3):178–82. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20732827
Geelhoed EA, Lewis B, Hounsome D, Leary PO. Economic evaluation of neonatal screening for phenylketonuria and congenital hypothyroidism. 2005;(April):575–9.
Lord J, Thomason MJ, Littlejohns P, Chalmers RA, Bain MD, Addison GM, et al. Secondary analysis of economic data : a review of cost-benefit studies of neonatal screening for phenylketonuria. 1999;179–86.
Carroll AE, Downs SM. Comprehensive Cost-Utility Analysis of Newborn. Pediatrics. 2006;
Venditti L, Venditti C, Berry G. Newborn screening by tandem mass spectrometry for medium-chain Acyl-CoA dehydrogenase deficiency: a cost-effectiveness analysis. Pediatrics [Internet]. 2003 [cited 2013 Jan 22]; Available from: http://pediatrics.aappublications.org/content/112/5/1005.short
Schoen E, Baker J, Colby C, To T. Cost-benefit analysis of universal tandem mass spectrometry for newborn screening. Pediatrics [Internet]. 2002 [cited 2013 Jan 22]; Available from: http://pediatrics.aappublications.org/content/110/4/781.short
Christianson A, Howson C, Modell B. March of Dimes Global Report on Birth Defects. 2006. 2006 [cited 2013 Jan 22]; Available from: http://www.marchofdimes.com/downloads/Birth_Defects_Report-PF.pdf

 Back to Blogs
Back to Blogs