- What is stem cell banking +
- Ordering Process +
- Pricing +
- Private vs Community +
- Uses for stem cells +
- FAQ +
- Downloads +
Safeguard your baby's future health
Stem cell banking is a form of medical insurance.
When your baby is born, their umbilical cord blood and tissue is saved so that it can be used in the future, potentially, to help in the treatment of a number of blood or immune disorders - should your child ever need it. To cryo-save refers to the process of collecting and preserving stem cells from your newborn's umbilical cord blood and/or tissue.
Like all insurance, you hope you never have to use it.
But if you do, you know that you have given your child the instant possibility of treatment, rather than adding their name to a donor waiting list.
What are stem cells?
We like to think of stem cells as the original building blocks of life.
They are the cells that make up the embryo and develop into different cell types in your body.
Umbilical cord blood and tissue is an excellent source of stem cells, because:
Saving them at birth is easy, painless and a lot less invasive than harvesting stem cells later in life. These stem cells are usually discarded as medical waste, which means that collecting them does not come with any moral, ethical or religious concerns. Scientists believe that treatment results are better when you use stem cells collected at birth because they are at the very beginning of their life span and do not show any age-related changes.

What is cord blood?
Your baby’s umbilical cord blood is high in blood-forming stem cells, which can be used to help treat over 80 blood-related diseases.
These stem cells are used in the regeneration of bone marrow as they can replace diseased or damaged cells with healthy new cells. They make it possible to rebuild a person’s blood and immune system.
How do you collect cord blood?
The umbilical cord is cut and clamped
The cord blood is collected by your Gynaecologist, Doctor or Midwife immediately after the birth of your baby, however you choose to give birth (vaginal birth or Caesarean section).
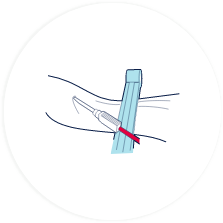
A needle is inserted into the umbilical cord vein and the blood is collected into a sterile bag.
The collection of the cord blood is quick, safe and painless for both mother and baby.
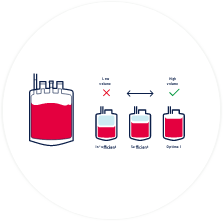
100 - 150ml of cord blood needs to be collected for it to be successfully stored.
If your Gynaecologist, Doctor or Midwife is not familiar with the process, we are happy to send them a video that takes them through the process in detail.
What is cord tissue?
Your baby’s umbilical cord tissue contains the type of stem cells which form the connective tissues in our bodies, such as skin, bone, muscle, cartilage, nerve and fat. These stem cells hold considerable potential in treating a wide variety of medical and aesthetic conditions, such as treating potential in treating burns or wounds that are battling to heal. They are currently being used in a number of clinical trials.
While these cells are found in most of our tissues or organs, they can be difficult to harvest. Collecting them at birth is best because:
The cells are "young" and have better regenerative potential.
Cord tissue is easily collected with no pain or discomfort to you or your child.
How do you collect cord tissue?

Cord tissue is collected by your Gynaecologist, Doctor or Midwife immediately after the birth of your baby.
After the cord blood has been collected, and after the placenta has been delivered, a 10 - 15cm piece of the umbilical cord is cut and cleaned.

The cord tissue is then placed into the collection tube and placed in the silver foil bag, along with the cord blood.
How are the stem cells stored?
After a pain-free collection process immediately following childbirth, the stem cells are transported to our specialised facility. There, they undergo rigorous processing and cryopreservation, a technique using ultra-low temperatures to maintain their viability for potential future medical use. The stem cells are stored in the vapour phase of liquid nitrogen in state-of-the-art storage tanks. The tanks are serviced annually by experts.
What does it mean when a sample is listed on the SABMR? When the stem cells are stored they will be tissue typed. This information is kept with the SABMR, which is used to determine if the stem cells are a match to a recipient in need. The stored stem cells will be used on a first come first serve basis. If your child / someone in your family requires the stem cells first, they will have full access to them. However, if the stem cells are found to be a match to someone else in need of a lifesaving transplant, the cells will be used by them. The parents will be informed that there is a match and will sign the consent for the stem cells to be given over, and Next Biosciences will then refund them the full amount paid to bank their baby’s sample with the Community Bank. Community Banking entry criteria:
Please note: The Family History Questionnaire needs to be completed prior to registering for Community Banking to assess your suitability for meeting the SABMR criteria. The questionnaire can be found in the downloads tab. Once completed, please send to netcells@nextbio.co.za and wait for feedback on your eligibility before registering. |
Did you know that your baby’s umbilical cord stem cells can be the lifeline to help treat over 80 blood-related diseases? And that umbilical cord tissue has the ability to differentiate into other specialised cell types – such as cartilage, bone, muscle, blood cells, skin and nerve cells? They provide a promising treatment alternative for the repair and regeneration of damaged tissues and organs. read more
We like to think of stem cells as the original building blocks of life. They are the cells that make up the embryo and develop into different cell types in your body. Saving them at birth is easy, painless and a lot less invasive than harvesting stem cells later in life.
On the African continent, Netcells is the longest-established stem cell banking facility. Based in South Africa and accredited by the American Association of Blood Banks (AABB), Netcells offers umbilical cord blood banking, as well as umbilical cord tissue banking.
Over the last 20 years, Netcells has banked over 23 000 units1 for parents who have chosen to protect and preserve the future health of their children, and we are now seeing their investment start to pay dividends.
Cord blood stem cells
Cord blood is a rich source of blood-forming stem cells (haematopoietic stem cells), which are already used in established medical treatments. These stem cells can rebuild the blood and immune system and form part of standard therapy for more than 80 conditions, including certain cancers, blood disorders, immune deficiencies, and metabolic diseases. read more
They are also being studied in ongoing clinical trials to explore their potential in treating additional conditions. While these emerging uses are still experimental, research continues to expand the future possibilities of cord blood stem cell therapy.
A few examples are:
Cord tissue stem cells
Recent developments have delivered some of the most exciting areas in research, particularly for mesenchymal stem cells (MSCs) from umbilical cord tissue. Globally, clinical trials are expanding with MSCs being investigated for their role in immune modulation, tissue repair, and regenerative therapies across fields such as orthopaedics, neurology, and autoimmune disease management. read more
Other sources of stem cells, such as adipose and bone marrow, decline in quantity and quality with age, and the ability to employ a patient’s own adult stem cells is not always feasible as cell quality may be impacted by specific disease or genetic conditions. MSCs from fetal origin have notably greater capacity to regenerate - this reinforces the growing importance of stored cord tissue and blood in treatment development.
2021 – 2023:
117 New clinical trials registered using cord-tissue derived MSCs2
1 622 Clinical trials registered utilising MSCs as a treatment regime3
Emerging applications
There has been significant and exciting progress in the potential uses of cord blood stem cells, both locally and internationally, with new studies exploring their application beyond traditional haematological conditions.
Recent Netcells-specific withdrawals
- Type I Diabetes – In March 2025, an umbilical cord blood unit was released for infusion in a 16-year-old with Type I Diabetes as part of an effort to assist in insulin regulation. Although the recipient continues to use insulin, their overall insulin requirements have significantly decreased. They also reported greater dietary flexibility without adverse effects and improved muscle mass, which was previously limited by their condition.
-
Autism Spectrum Disorder (ASD) – In April and July 2025, two units of cultured stem
cells from autologous (self-derived) umbilical cord tissue were released. These were successfully
transplanted to support therapeutic management of symptoms associated with ASD.
- Patient 1 (Aphasic autism) – Prior to treatment, expressive communication was largely limited to single word responses. Two months post-transplant, the recipient demonstrated marked improvement, even participating in a school spelling bee.
- Patient 2 – This recipient is exhibiting improved calmness, engagement and responsiveness after transplantation of umbilical cord stem cells.
Netcells has a number of anticipated releases of cultured MSCs coming up, one of which is for the treatment and management of severe neck pain in an elderly family member, which previous surgeries have not managed to alleviate.
International withdrawals by SmartCells
(Netcells’ sister bank with facilities in the UK and Dubai)
SmartCells is part of the FamiCord group – the largest cord blood bank in Europe.
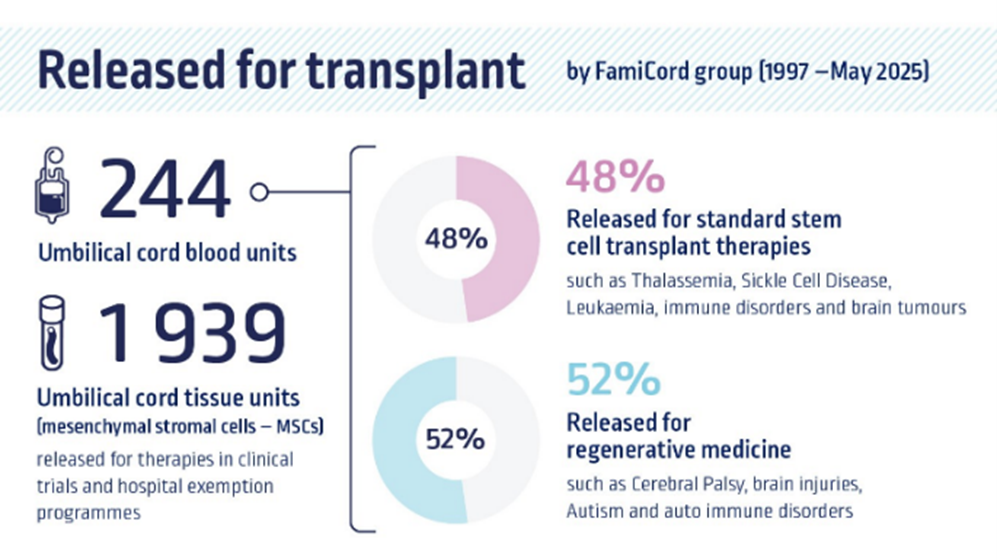
To-date, 2444 umbilical cord blood units and 1 939 umbilical cord tissue units have been released for transplant.
Get a Netcells quote today, and find out more about our storage options and personalised pricing:
Email: netcells@nextbio.co.za
Call: 011 697 2900 or 084 664 4646
WhatsApp: 084 664 4646
References:
- https://nextbio.co.za/Portals/0/adam/SharedDocuments/newsletters/netcells-parent-newsletter-2025-q4.pdf
- https://parentsguidecordblood.org/en/news/famicord-europes-largest-cord-blood-bank-and-largest-manufacturer-cord-tissue-msc-therapy
- https://cells4life.com/2024/09/key-takeaways-from-umbilical-cord-stem-cell-trial-research/
- https://nextbio.co.za/blog/details/where-does-cord-blood-go-global-trends-in-wit-2
What are the steps if I would like to bank my baby’s stem cells?
- Register on-line via the easy-to-use on-line registration platform and make payment for the registration fee.
- We will deliver a collection kit to you and one of our client services team will take you through the process to ensure you are properly informed.
- You take the collection kit with you to the hospital for your Gynae/Midwife to do the collection.
Who can make use of the stem cells?
The stem cells are a perfect match for the baby that they were collected for. There is also a 25% chance that they will be a match for a sibling. As the stem cells belong to the family, they are stored exclusively for their use.
Why should I bank my baby's stem cells?
- Saving them at birth is easy, painless and a lot less invasive than harvesting stem cells later in life.
- Your chances of finding a stem cell donor match are 1 in 100000 and the odds drop much further for mixed race children or ethnicities that are underrepresented on stem cell registries.
- The cord blood stem cells will be a perfect match for your baby, which means no risk of rejection.
- Stem cells collected at birth are biologically younger than stem cells collected later in life and have better regenerative potential.
Are there instances where the stem cells collected could not be used?
If the blood disorder is hereditary, the cord blood stem cells could not be used for treatment as the disease is found in the stem cells as well. In these situations, you would look to use a sibling’s stem cells, should they be a tissue match.
Are there situations where the cord blood cannot be collected, what happens then?
There can be instances in which the cord blood cannot be collected, which can occur if there are any difficulties with the labour in which your gynae/midwife needs to prioritise the health and safety of mom and/or the baby over collecting stem cells. Should this happen, you will be refunded the processing and storage fees that you have paid.
Situations in labour / birth that can cause low volume or no collection:
- Placental Abruption
- Placenta praevia
- Eclampsia/ pre-eclampsia
- Patients request for delayed cord clamping
- Maternal or foetal distress
- Premature deliveries
- Multiple births
- Small low birth weight babies / small placenta/ calcified placenta
- Thin umbilical cord that makes milking of the cord difficult
- Maternal health conditions such as hypertension, diabetes
Is anyone eligible to bank with the community bank?
There are eligibility criteria for the community bank. This is because the South African Bone Marrow Registry needs to ensure that the cord blood sample is not potentially affected by anything which may be transferred over to a recipient in need during a stem cell transplant.
We need to look at the biological mother and fathers’ and both their families medical history to determine your risk for passing on a blood transmissible disease or an inherited blood, autoimmune, metabolic or neurological disorder to your child which may affect their cord blood
We will screen the mother for any blood transmissible viruses she could have passed on to her child during pregnancy.
If I am having twins/triplets do I need to store for each baby?
We recommend that cord blood/ cord tissue is banked separately for each child in the case of twins or triplets, however individual cases can be discussed with our Medical Director.
Non-identical twins and non-identical triplets are conceived from different eggs and sperm and are no more alike than siblings born at separate times. The chance of a tissue type (HLA match) between siblings is 25%. In these cases, it is advised to store for each baby as their tissue types are different. Having two units stored increases the likelihood of a useful match between family members and it is possible to combine smaller units should siblings be a match to ensure an adequate stem cell yield for transplantation if ever needed.
For identical twins or rarely identical triplets the fertilized egg divides at an early stage of development and the babies will have the same genetic information. However even in these cases, minor genetic changes may occur after conception and during foetal development. While identical twins are genetically identical, small genetic differences may exist at varying stages of development, causing a low but present risk of differences in disease profiles. For example, one twin may develop leukaemia or diabetes and the other does not.
However, if you request only one collection for cord blood and tissue for identical twins, it is possible to do so after discussing this with our Medical Director. Each case should be discussed with the Medical Director and your obstetrician and the implications of this explained to them. The obstetrician must be sure that this is a case of identical twins for this to be considered.
Either a single collection can be done from one twin with the option to use the unit for the other twin should the need arise OR cord blood can be collected from each umbilical cord (in the case of a single placenta) and combined in one bag. This will lead to a situation of two genetically identical donors for a single unit and hence we recommend that in these cases the unit should be used for syngeneic transplant (i.e. a stem cell transplant only between the identical twins) and possibly not for other siblings. However, should one baby develop a genetic disorder requiring a transplant and the blood is combined, the transplant doctor may decide not to use the cord blood stem cells as they may contain the same genetic problem.
Cord tissue does not require as stringent HLA matching as cord blood so it is possible to only store one umbilical cord tissue unit for both babies.
Please note that collections for any multiple pregnancy has a higher risk of a lower volume with lower cell counts. This is because multiple pregnancies can often be premature, with smaller babies with lower birth weights and smaller placentas. In the case of emergencies where the mother’s health or that is of the babies is of utmost importance, the doctor may decide to abandon the cord blood collection procedure and safely deliver the babies.
What are the odds of using cord blood stem cells?
Ideally, your child or their sibling’s stem cells will never be needed and should be regarded as an extra form of medical insurance.
Studies in the USA have been done to calculate a lifetime probability (age 0-70) that a person will undergo a stem cell transplant:
1 in 435 people may receive their own stem cells
1 in 400 people will receive someone else’s cells
The total combined number of stem cell transplants is 1 in 217 people.
As more clinical uses become evident, these statistics are likely to increase the likelihood of use particularly for more commonly occurring medical illnesses.
What happens if I have paid in full and the cord blood is unable to be stored?
Next Biosciences will always refund the portion of the fee relating to the service that wasn’t utilized. Additionally, Next Biosciences takes the risk on processing, so if for whatever reason there are not enough stem cells for storage and you don’t store, then you will refund the banking and storage fees.
Can one family donate their stored cord blood stem cells to family and friends?
Cord blood stored with the private bank belongs to that family – the parents are the guardians of the cord blood until the child is 18 years old. They are entitled to store the cord blood for however long they like, and they are entitled to donate it to whoever they like. If it is found that a friend’s child needs a stem cell transplant and that their child’s stem cells are a match, they are allowed to donate the stored stem cells if they wish. They are not allowed to receive remuneration for the donation. South Africa has laws prohibiting the sales of organs, blood etc.
Before releasing stem cells, Netcells will insist on documentation from the transplanting doctor. If this is found to be legitimate and the stem cells are going to a registered doctor/ hospital/ institution, we will not have a problem releasing them to the transplanting clinic.
What is the procedure if the cells are needed?
The cord blood stem cells are ready for use. The administering doctor will contact us, provide the relevant documentation and we will co-ordinate the transport of the cells to them.
Cord tissue stem cells will need to be cultured to whatever use they are needed for e.g. bone regeneration or skin regeneration etc. Those protocols are still in development and another laboratory who specialise in this may need to be used. But that is all to be assessed in the future.
What are the retrieval costs for cord blood and cord tissue?
Cord blood – there are no retrieval costs. The stem cells are ready to be used.
Cord tissue – the cells need to be extracted from the tissue and cultured. The cells may then need to be manipulated further depending on the treatment required. There will be a charge for this process, but it is unknown as treatments have not been developed yet.
Cost of therapy – cord blood transplantation for bone marrow regeneration is generally covered by medical aid.
Finding a match – through the bone marrow registry is approx. R250 000.
Cost of buying the stem cells from overseas – anywhere between R300000 and R1million.
Has Netcells released any stored units for treatment?
Netcells has released 3 stem cells units.
We released our first cord blood unit in March 2015 to treat an eight-month-old baby boy at Duke University in the USA.
We released our second cord blood unit in June 2016 to treat a 3-year-old boy with cerebral palsy at Duke University.
We have had a few siblings who required stem cell transplants but these were unfortunately not a match. Our sister company Smart Cells in the UK has already released 21 cord blood units for the treatment for cerebral palsy, thalassaemia, leukaemia, brain injury, severe combined immunodeficiency disorder, sickle cell anaemia and encephalitis.
How many stem cells are needed for transplantation?
This depends on the number of stem cells stored in each individual unit and the size of the person who requires the transplant. The optimal dose is 10 -20 million nucleated (CD45) cells per kilogram of body weight. The cell counts are different for every collection and depend on the volume of blood collected as well as other factors in pregnancy. Your baby’s cell counts will be reported to you on your storage certificate.
The average number of stem cells collected from an average volume of 100 ml can usually treat up to a 50kg person, but this varies. Some of the units contain large numbers of cells which would be enough to treat adults with a single unit and others are much smaller and are suitable for smaller children. In larger adults, often two matched units can be combined (either from a sibling or an unrelated donor).
Companies are developing techniques to expand (grow more) stem cells in a laboratory environment, and we hope this will become a reality soon.
It must be noted that lower stem cell counts are currently being used in clinical trials for cerebral palsy and brain injury, as these recipients are often small, low birth weight babies and children.
Why should I renew my storage with Netcells?
Keeping your child’s stored cord blood and tissue can continue to provide peace of mind knowing you have a shield against life’s uncertainties. Cells derived from the cord blood and cord tissue continue to have a fundamental advantage to treat disease than cells derived later in life and are a valuable resource to continue to invest in should you ever need regenerative medicine procedures or disease treatment in the future. Stem cell research continues to evolve, and new treatments are emerging. Renewing storage ensures access to these future therapies.

Enter your details to find out more about how stem cell banking can positively impact your family's health.
Thank you for contacting us. We will respond within one working day.
Next Biosciences
Ariane Avenue
International Business Gateway
Cnr. New Road and 6th Road
Midrand, South Africa
Office Hours
Monday - Thursday: 08h00 - 17h00
Friday: 08h00 - 16h00